Membrane Attack Complex (MAC): The Complement Cascade's Final Punch
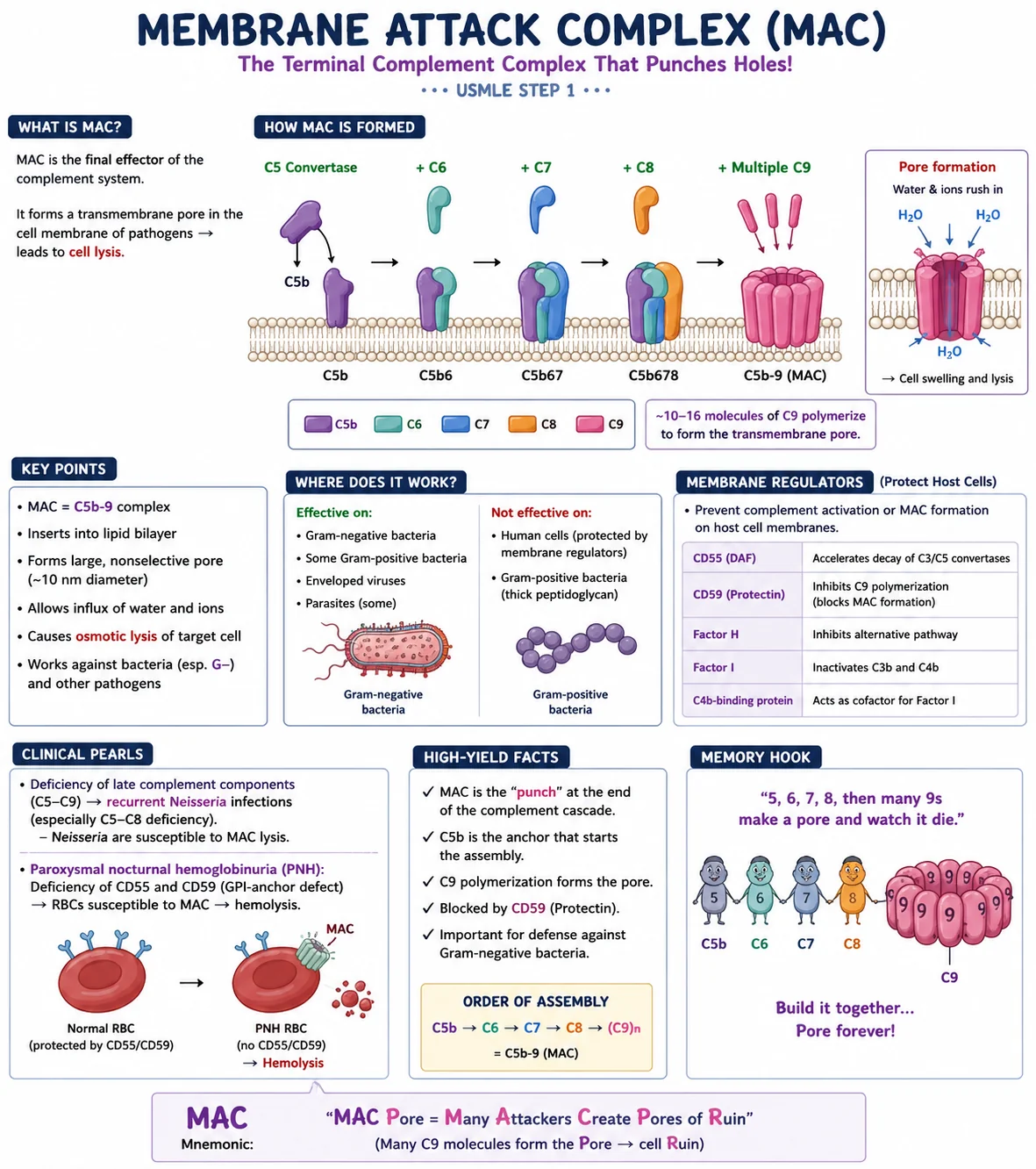
Quick summary
- What it is: the C5b-9 transmembrane pore — the final step of the complement cascade.
- How it forms: C5 convertase cleaves C5 → C5b binds the membrane → C6, C7, C8 join → ~10–16 C9 molecules polymerize → ring-shaped pore (~10 nm).
- What it kills: Gram-negative bacteria (especially Neisseria), enveloped viruses, some parasites; ineffective on Gram-positive bacteria (thick peptidoglycan) and host cells (CD55/CD59 protect).
- High-yield diseases: late complement deficiencies (C5–C9) → recurrent Neisseria infections; PNH (CD55/CD59 loss) → RBC hemolysis.
Memory hook
5, 6, 7, 8, then many 9s — make a pore and watch it die. Build it together → pore forever.
What the membrane attack complex actually does
The complement system has three activation pathways (classical, lectin, alternative), but they all converge on the same finale: a transmembrane pore that punches a hole in the target cell. That pore is the membrane attack complex (MAC), also known as C5b-9. Water and ions rush in along their gradients, the cell swells, and osmotic lysis kills it.
MAC is the "punch" at the end of the complement cascade. Everything upstream — opsonization, anaphylatoxins, chemotaxis — supports either flagging the target or recruiting the cells that build the MAC.
How MAC assembles, step by step
The pore is built sequentially on the target membrane:
- C5 convertase cleaves C5 into C5a (anaphylatoxin) and C5b.
- C5b anchors to the target membrane and recruits the next component.
- +C6 joins to form C5b6.
- +C7 binds; the complex inserts into the lipid bilayer (C5b67).
- +C8 binds; partial pore begins to form (C5b678).
- ~10–16 C9 molecules polymerize around the C5b-8 platform, forming a ring-shaped, water-filled transmembrane pore — the completed C5b-9 (MAC).
The pore is large (~10 nm), nonselective, and irreversible. Water and ions equilibrate across the membrane, the cell swells, and osmotic lysis kills it.
Where MAC works (and where it doesn't)
Effective targets:
- Gram-negative bacteria — thin peptidoglycan layer; MAC reaches the inner membrane. Most clinically important.
- Some Gram-positive bacteria.
- Enveloped viruses.
- Some parasites.
Resistant or protected:
- Gram-positive bacteria — the thick peptidoglycan cell wall sterically blocks MAC insertion.
- Host cells — protected by membrane-bound complement regulators (CD55, CD59).
| Target | MAC effective? | Why |
|---|---|---|
| Neisseria (Gram-) | Yes | Thin peptidoglycan; MAC reaches inner membrane |
| Most Gram-positive bacteria | No | Thick peptidoglycan blocks insertion |
| Enveloped viruses | Yes | Lipid envelope is vulnerable to pore |
| Host RBCs (normal) | No | CD55/CD59 prevent MAC on host membranes |
| PNH RBCs | Yes | CD55/CD59 absent → hemolysis |
Membrane regulators that protect host cells
Host cells express several proteins that prevent MAC from forming on their own membranes. These are critical to know:
- CD55 (Decay-Accelerating Factor, DAF) — accelerates the decay of C3 and C5 convertases, so less C5b is generated on host cells.
- CD59 (Protectin) — directly inhibits C9 polymerization, blocking MAC formation. The terminal blocker.
- Factor H — inhibits the alternative pathway by competing for C3b binding.
- Factor I — serine protease that inactivates C3b and C4b.
- C4b-binding protein — cofactor for Factor I, enhances C4b inactivation.
CD55 and CD59 are GPI-anchored to the cell membrane. Their absence is the molecular basis of paroxysmal nocturnal hemoglobinuria.
High-yield clinical pearls
Late complement deficiencies → recurrent Neisseria
A deficiency of any terminal complement component (C5, C6, C7, C8, or C9 — and the regulator Properdin) impairs MAC formation. Neisseria species (N. meningitidis, N. gonorrhoeae) are uniquely susceptible to MAC-mediated lysis because of their thin peptidoglycan, so patients with terminal complement defects present with recurrent disseminated Neisserial infections — meningococcemia, gonococcal arthritis, recurrent meningitis.
Buzzword: recurrent Neisseria infections on a USMLE vignette → think late complement (C5–C9) deficiency. Order a CH50 — it'll be low (CH50 measures the entire classical pathway through MAC).
Paroxysmal nocturnal hemoglobinuria (PNH)
PNH is caused by an acquired mutation in the PIGA gene in a hematopoietic stem cell. PIGA encodes an enzyme required to make GPI anchors. Without GPI anchors, CD55 and CD59 cannot be tethered to the cell membrane.
Result: the patient's RBCs (and other cells from the affected clone) lack CD55/CD59 protection, so spontaneous low-grade complement activation generates MAC on RBC membranes → chronic intravascular hemolysis. Classic features:
- Hemoglobinuria, often most prominent in the morning ("paroxysmal nocturnal").
- Pancytopenia (bone marrow stem-cell defect).
- Venous thrombosis (especially hepatic, mesenteric, cerebral) — a major cause of death.
- Treated with eculizumab (anti-C5 monoclonal antibody, blocks C5 cleavage and therefore MAC formation).
PNH is the canonical "complement regulator deficiency" vignette.
Atypical hemolytic uremic syndrome (aHUS)
A useful contrast: aHUS is caused by deficiency of Factor H, Factor I, or MCP (CD46) — the alternative-pathway regulators. Uncontrolled complement activation on endothelium causes microangiopathic hemolytic anemia + acute kidney injury. Also treated with eculizumab.
High-yield facts (board-ready)
- MAC is the C5b-9 complex — the "punch" at the end of the complement cascade.
- C5b is the anchor that starts assembly.
- C9 polymerization forms the pore (~10–16 C9 molecules).
- CD59 (Protectin) is the host's terminal MAC blocker.
- Important against Gram-negative bacteria — especially Neisseria.
- Recurrent Neisseria = late complement (C5–C9) deficiency.
- PNH = loss of CD55 + CD59 → MAC-mediated hemolysis.
Frequently asked questions
What is the membrane attack complex (MAC)?
The MAC is the C5b-9 complex — the final effector of the complement system. It's a transmembrane pore that forms in the membrane of pathogens, allowing water and ions to flood in, leading to osmotic cell lysis. It's the "killing" step at the end of all three complement activation pathways (classical, lectin, alternative).
How is the membrane attack complex formed?
C5 convertase cleaves C5 into C5a and C5b. C5b anchors to the target membrane and sequentially recruits C6, C7, and C8. Then ~10 to 16 molecules of C9 polymerize to form a ring-shaped transmembrane pore (~10 nm in diameter). The completed assembly is called C5b-9 or the membrane attack complex.
Why does CD59 deficiency cause hemolysis?
CD59 (Protectin) blocks C9 polymerization on host cell membranes. Without CD59, spontaneous low-grade complement activation generates MAC on host RBCs, causing intravascular hemolysis. This is the molecular basis of paroxysmal nocturnal hemoglobinuria (PNH), where CD59 (and CD55) cannot be GPI-anchored to the membrane because of a PIGA mutation.
Why are patients with C5–C9 deficiency prone to Neisseria infections?
Neisseria species (N. meningitidis and N. gonorrhoeae) are Gram-negative bacteria with thin peptidoglycan, making them uniquely susceptible to MAC-mediated lysis. Without functional MAC (due to C5–C9 deficiency), the immune system loses its primary mechanism for killing Neisseria, leading to recurrent disseminated infections like meningococcemia and gonococcal arthritis.
Which complement deficiency causes paroxysmal nocturnal hemoglobinuria?
PNH isn't a deficiency of a complement protein itself — it's a deficiency of the regulators CD55 and CD59 caused by an acquired PIGA gene mutation. Without GPI anchors, these regulators can't attach to the cell membrane, so MAC forms on RBCs and causes intravascular hemolysis. PNH is treated with eculizumab, an anti-C5 monoclonal antibody that prevents MAC formation upstream.
What's the difference between CD55 and CD59?
CD55 (DAF) accelerates decay of C3 and C5 convertases, preventing the cascade from reaching the MAC step. CD59 (Protectin) directly blocks C9 polymerization, preventing the final pore from forming. CD55 acts upstream; CD59 is the terminal blocker. Both are GPI-anchored and both are deficient in PNH.
Why doesn't MAC kill Gram-positive bacteria?
Gram-positive bacteria have a thick peptidoglycan layer outside the cell membrane. The MAC physically can't insert into the inner membrane through that wall. This is why complement is most clinically critical against Gram-negative pathogens — and why the late-complement-deficiency vignette is always Neisseria, not Streptococcus or Staphylococcus.
Educational use only. This article is a study reference for medical students preparing for the USMLE. It is not medical advice and is not a substitute for evaluation by a qualified clinician.
Try Noteflix free